property>critical point temperature
What is the Critical Temperature?
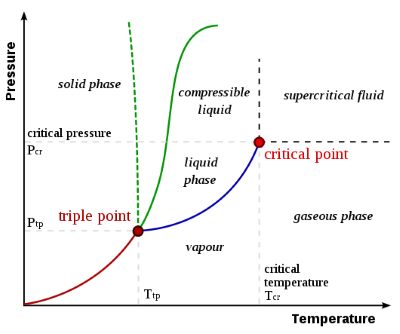
The critical temperature (Tc, or critical point temperature) is the temperature above which a gas cannot be made to liquefy, no matter how great the pressure applied. The corresponding critical pressure (Pc) is the minimum pressure that must be applied to bring about liquefaction at the critical temperature.
The Gas-Liquid Critical Point
The critical point temperature or critical state temperature of any material is that temperature above which there is no distinct existence of liquid and gas phases. The phase properties of the gas and liquid become the same as the critical point temperature approaches. Only one phase exists at this point which is also known as a homogeneous supercritical fluid when the temperature is equal to the critical point temperature. The heat of vaporization becomes zero at this critical point temperature. No further increase in pressure can form a liquid but solidification may occur if the pressure is increased sufficiently. The critical point temperature is a property of the substance. For water, the critical point occurs at around 647 K (374 °C or 705 °F) and 22.064 MPa (3200 PSIA or 218 atm).
Other Related Properties
The critical molar volume represents the volume of one mole of substance at the critical temperature and pressure. The liquid-liquid critical point of a solution is the point at which it gets separated into two different liquids of different densities. This is the point at which any change in temperature or pressure will separate the mixture into two distinct liquid phases. Such points are also known as the critical solution temperature. The upper critical solution temperature (UCST) is the warmest point at which cooling will cause phase separation, and the lower critical solution temperature (LCST) is the coolest point at which heating will cause phase separation.
Divergences at Critical Point
Every fluid has a different critical point. The end of the vapour pressure is marked by the critical point on the pressure-temperature curve; this phase transition is different from the phenomenon of boiling that occurs along the vapor pressure curve. There are other anomalies near the critical point as the isothermal compressibility and heat capacity approach infinity as the fluid approaches Tc.
The critical points of ferromagnetic materials such as iron are known as the Curie Point, where, again, a number of anomalies occur including singularities in heat capacity and divergent magnetic properties.